21 CFR Part 11 Compliance for Spreadsheets Overview
CIMCON's Document Control Software is essential for pharmaceutical and biotech companies needing to comply with 21 CFR Part 11 regulations. This software facilitates digital transformation by ensuring precise document management and control. It is particularly valuable for teams managing compliance documentation, offering features like e-signatures and training on critical documents and data. Users can digitize existing forms and maintain data integrity in lab systems, which is crucial for audit readiness and risk assessments. Known for its reliability in regulatory environments, CIMCON supports quality assurance managers in maintaining compliance and avoiding costly FDA warning letters.
Use Cases
Customers recommend Digital Signature, Workflow Management, Competitive Intelligence, as the business use cases that they have been most satisfied with while using 21 CFR Part 11 Compliance for Spreadsheets.
Business Priorities
Enhance Customer Relationships and Scale Best Practices are the most popular business priorities that customers and associates have achieved using 21 CFR Part 11 Compliance for Spreadsheets.
21 CFR Part 11 Compliance for Spreadsheets Use-Cases and Business Priorities: Customer Satisfaction Data
21 CFR Part 11 Compliance for Spreadsheets's features include Recording. and 21 CFR Part 11 Compliance for Spreadsheets support capabilities include 24/7 Support, Chat Support, Email Support, etc. also 21 CFR Part 11 Compliance for Spreadsheets analytics capabilities include Custom Reports, and Analytics.
Reviews
"...Being an Infrastructure company with less tech savvy end-users, we could easily roll the solution in 2 months time with 100+ users...." Peer review by Jayanta N., Administrator
21 CFR Part 11 Compliance for Spreadsheets, belong to a category of solutions that help CPQ. Each of them excels in different abilities. Therefore, determining the best platform for your business will depend on your specific needs and requirements.
21 CFR Part 11 compliance is ensured by the eInfotree Excel Module from CIMCON Software. Data management is revolutionized with its seamless integration and regulatory adherence.
21 CFR Part 11 Compliance for Spreadsheets Customer wins, Customer success stories, Case studies
What makes 21 CFR Part 11 Compliance for Spreadsheets ideal for Digital Signature?
How can 21 CFR Part 11 Compliance for Spreadsheets enhance your Workflow Management process?
11 buyers and buying teams have used Cuspera to assess how well 21 CFR Part 11 Compliance for Spreadsheets solved their CPQ needs. Cuspera uses 204 insights from these buyers along with peer reviews, customer case studies, testimonials, expert blogs and vendor provided installation data to help you assess the fit for your specific CPQ needs.
21 CFR Part 11 Compliance for Spreadsheets Features
- Low
- Medium
- High
| FEATURE | RATINGS AND REVIEWS |
|---|---|
| Custom Reports | Read Reviews (13) |
| Analytics | Read Reviews (4) |
| CAPABILITIES | RATINGS AND REVIEWS |
|---|---|
| Custom Reports | Read Reviews (13) |
| Analytics | Read Reviews (4) |
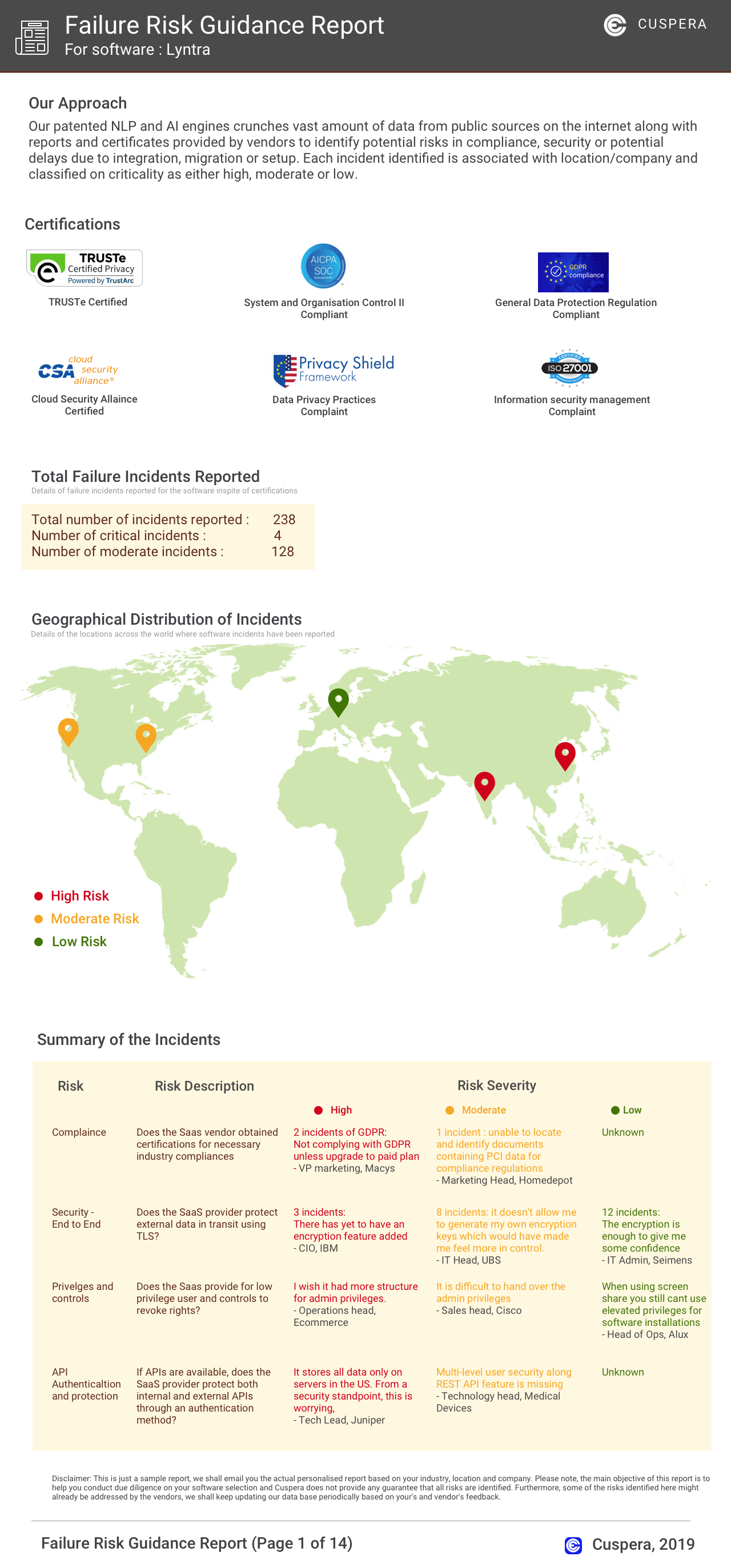
Software Failure Risk Guidance
?for 21 CFR Part 11 Compliance for Spreadsheets
Overall Risk Meter
Top Failure Risks for 21 CFR Part 11 Compliance for Spreadsheets
CIMCON Software Profile
Company Name
CIMCON Software
Company Website
https://part11solutions.com/HQ Location
234 Littleton Road,,Westford, MA 01886, USA
Employees
11-50
Social