Certara GlobalSubmit eCTD software Overview
Globalsubmit provides eCTD submission software to mitigate rejection risks in a complex regulatory environment. Differences in regulatory requirements complicate the submission process.
Use Cases
Customers recommend Sending & Publishing Communications, Workflow Management, Onboarding, as the business use cases that they have been most satisfied with while using Certara GlobalSubmit eCTD software.
Business Priorities
Scale Best Practices and Launch New Products are the most popular business priorities that customers and associates have achieved using Certara GlobalSubmit eCTD software.
Certara GlobalSubmit eCTD software Use-Cases and Business Priorities: Customer Satisfaction Data
Certara GlobalSubmit eCTD software's features include Templates. and Certara GlobalSubmit eCTD software support capabilities include 24/7 Support, Email Support, Chat Support, etc. also Certara GlobalSubmit eCTD software analytics capabilities include Custom Reports, and Analytics.
Reviews
"...GlobalSubmits approach to document management is a familiar one intuitive interface, easy implementation that appeals to the novice user, built-in templates and workflows, and integration with eCTD publishing systems...." Peer review
Peer review evidence (same sources as the product rating summary)
"...PUBLISH is a mature product that is now the second most used solution for eCTD publishing...." Regulatory Submissions Software
"...user workflow by cutting out at least four publishing tasks per document...." PUBLISH
"...Installation and user guides, onboarding and training, including manual practice documents and individualized Q&A session...." Authoring Templates
Popular Business Setting
for Certara GlobalSubmit eCTD software
Top Industries
- Pharmaceuticals
Popular in
- Enterprise
Certara GlobalSubmit eCTD software is popular in Pharmaceuticals, and is widely used by Enterprise,
Certara GlobalSubmit eCTD software Customer wins, Customer success stories, Case studies
How does Certara GlobalSubmit eCTD software address your Sending & Publishing Communications Challenges?
How can Certara GlobalSubmit eCTD software optimize your Workflow Management Workflow?
What solutions does Certara GlobalSubmit eCTD software provide for Onboarding?
How does Certara GlobalSubmit eCTD software address your Content Management Challenges?
11 buyers and buying teams have used Cuspera to assess how well Certara GlobalSubmit eCTD software solved their business needs. Cuspera uses 376 insights from these buyers along with peer reviews, customer case studies, testimonials, expert blogs and vendor provided installation data to help you assess the fit for your specific business needs.
Certara GlobalSubmit eCTD software Features
- Low
- Medium
- High
| FEATURE | RATINGS AND REVIEWS |
|---|---|
| Custom Reports | Read Reviews (48) |
| Analytics | Read Reviews (1) |
| CAPABILITIES | RATINGS AND REVIEWS |
|---|---|
| Custom Reports | Read Reviews (48) |
| Analytics | Read Reviews (1) |
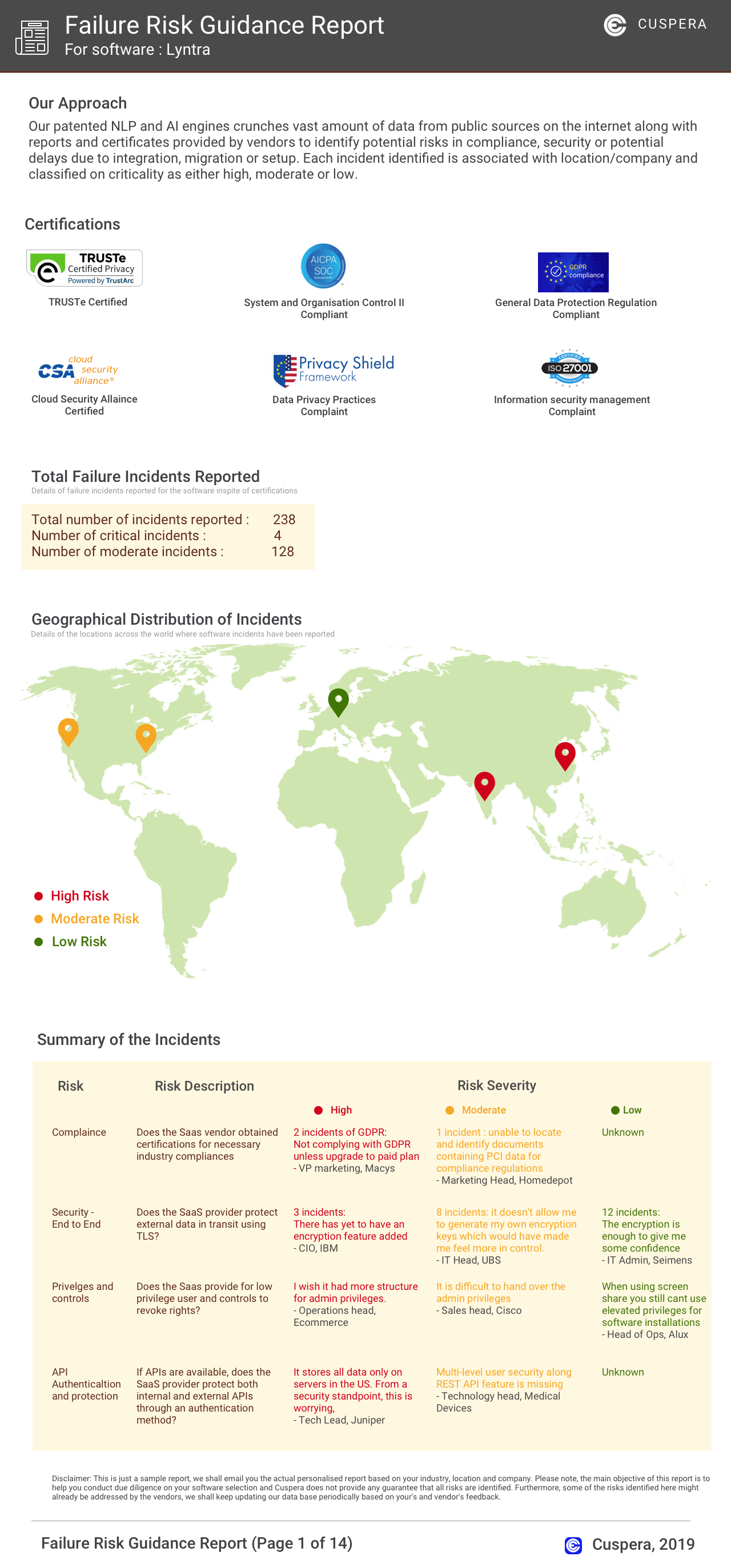
Software Failure Risk Guidance
?for Certara GlobalSubmit eCTD software
Overall Risk Meter
Top Failure Risks for Certara GlobalSubmit eCTD software
Certara, USA. News
Certara stock (US1567671046): Q1 revenue up 1% but services decline sparks investigation
Certara reported a 1% increase in Q1 2026 revenue to $106.9 million, despite a 4% decline in services revenue. This led to a securities fraud investigation by Kirby McInerney LLP. The company's core biosimulation software remains crucial for drug development, supporting major pharma firms like Pfizer and Moderna.
Certara Q1 2026 results: sells medical writing unit for $85M
Certara reported Q1 2026 revenue of $106.9 million, with software revenue up 7% and services revenue down 4%. The company sold its medical writing unit to Veristat for $85 million, aiming to focus on drug development. Certara updated its 2026 revenue guidance to $395-$405 million.
Certara Reports First Quarter 2026 Financial Results
Certara reported its Q1 2026 financial results, showing a 1% revenue increase to $106.9 million. Software revenue rose 7% to $49.7 million, while services revenue decreased by 4% to $57.2 million. The company posted a net loss of $8.8 million, compared to a net income of $4.7 million the previous year. Certara completed the sale of its medical writing business to Veristat for up to $135 million.
Altasciences and Certara Announce Strategic Partnership to Accelerate ...
Certara and Altasciences have formed a strategic partnership to enhance early-phase drug development. By integrating Certara's biosimulation technology with Altasciences' Acceleration Platform, the collaboration aims to improve study designs, refine dosing strategies, and enable faster, informed decision-making. This model-first approach supports adaptive, data-driven drug development, aligning with FDA guidance and benefiting biotech sponsors and patients globally.
Certara, USA. Profile
Company Name
Certara, USA.
Company Website
https://www.certara.com/HQ Location
123 South Broad Street, Suite 1850, Philadelphia, PA 19109, US
Employees
11-50
Social
Financials
PRIVATE